Unbounding the Future:
the Nanotechnology Revolution
Chapter 2
The Molecular World
Nanotechnology
will be a bottom-up technology, building upward from the
molecular scale. It will bring a revolution in human abilities
like that brought by agriculture or power machinery. It can even
be used to reverse many of the changes brought by agriculture or
power machinery. But we humans are huge creations with no direct
experience of the molecular world, and this can make
nanotechnology hard to visualize, hence hard to understand.
Scientists working with moleculas face this
problem today. They can often calculate how molecules will behave, but to
understand this behavior, they need more than heaps of
numbers: they need pictures, movies, and interactive simulations,
and so they are producing them at an ever-increasing pace. The
U.S. National Science Foundation has launched a programm in
"scientific visualization", in part to harness
supercomputers to the problem of picturing the molecular world.
Molecules are objects that exert forces on one
another. If your hands were small enough, you could grab them,
squeeze them, and bash them together. Understanding the molecular
world is much like understanding any other physical world: it is
a matter of understanding size, shape, strength, force, motion,
and the like–a matter of understanding the differences
between sand, water, and rock, or between steel and soap bubbles.
Today's visualization tools give a taste of what will become
possible with tomorrow's faster computers and better
"virtual realities," simulated environments that let
you tour a world that "exists" only as a model inside
the computer. Before discussing nanotechnology and how it relates
to the technologies of today, let's try to get a more concrete
understanding of the molecular world by describing a simulation
embedded in a scenario. In this scenario, events and technologies
described as dating from 1990 or before are historically
accurate; those with later dates are either projections or mere
scenario elements. The descriptive details in the simulation are
written to fit designs and calculations based on standard
scientific data, so the science isn't fiction.
Exploring the Molecular
World
In a scenario in the last chapter, we saw Joel
Gregory manipulating molecules in the virtual reality of a
simulated world using video goggles, tactile gloves, and a
supercomputer. The early twenty-first century should be able to
do even better. Imagine, then, that today you were to take a
really long nap, oversleep, and wake up decades later in a
nanotechnological world.
In the twenty-first century, even more than
in the twentieth, it's easy to make things work without
understanding them, but to a newcomer much of the technology
seems like magic, which is dissatisfying. After a few days,
you want to understand what nanotechnology is, on a gut
level. Back in the late twentieth century, most teaching used
dry words and simple pictures, but now—for a topic like
this—it's easier to explore a simulated world. And so
you decide to explore a simulation of the molecular world.
Looking through the brochure, you read many
tedious facts about the simulation: how accurate it is in
describing sizes, forces, motions, and the like; how similar
it is to working tools used by both engineering students and
professionals; how you can buy one for your very own home,
and so forth. It explains how you can tour the human body,
see state-of-the-art nanotechnology in action, climb a
bacterium, etc. For starters, you decide to take an
introductory tour: simulations of real twentieth-century
objects alongside quaint twentieth-century concepts of
nanotechnology.
After paying a small fee and memorizing a few
key phrases (any variation of "Get me out of here!"
will do the most important job), you pull on a powersuit,
pocket a Talking Tourguide, step into the simulation chamber,
and strap the video goggles over your eyes. Looking through
the goggles, you seem to be in a room with a table you know
isn't really there and walls that seem too far away to fit in
the simulation chamber. But trickery with a treadmill floor
makes the walk to the walls seem far enough, and when you
walk back and thump the table, it feels solid because the
powersuit stops your hand sharply at just the right place.
You can even feel the texture of the carvings on the table
leg, because the suit's gloves press against your fingertips
in the right patterns as you move. The simulation isn't
perfect, but it's easy to ignore the defects. On the table is
(or seems to be) an old 1990s silicon computer chip. When you
pick it up, as the beginners' instructions suggest, it looks
like Figure 1A. Then you say, "Shrink me!", and the
world seems to expand.
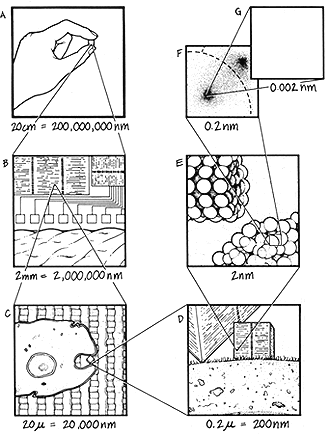
FIGURE 1: POWER OF TEN
Frame (A) shows a hand holding a computer chip. This is
shown magnified 100 times in (B). Another factor of 100
magnification (C) shows a living cell placed on the chip to
show scale. Yet another factor of 100 magnification (D)
shows two nanocomputers
beside the cell. The smaller (shown as block) has roughly
the same power as the chip seen in the first view; the
larger (with only the corner visible) is as powerful as
mid-1980s mainframe computer. Another factor of 100
magnification (E) shows an irregular protein from the
cell on the lower right, and a cylindrical gear made by molecular
manufacturing at top left. Taking a smaller factor of
10 jump, (F) shows two atoms in the protein, with
electron clouds represented by stippling. A final factor
of 100 magnification (G) reveals the nucleus of the atom
as a tiny speck.
Vision and Motion
You feel as though you're falling toward the
chip's surface, shrinking rapidly. In a moment, it looks
roughly like Figure 1B, with your thumb still there holding
it. The world grows blurrier, then everything seems to go
wrong as you approach the molecular level. First, your vision
blurs to uselessness—there is light, but it becomes a
featureless fog. Your skin is tickled by small impacts, then
battered by what feel like hard-thrown marbles. Your arms and
legs feel as though they are caught in turbulence, pulling to
and fro, harder and harder. The ground hits your feet, you
stumble and stick to the ground like a fly on flypaper,
battered so hard that it almost hurts. You asked for realism,
and only the built-in safety limits in the suit keep the
simulated thermal motions of air molecules and of your own
arms from beating you senseless.
"Stop!" gives you a rest from the
suit's yanking and thumping, and "Standard
settings!" makes the world around you become more
reasonable. The simulation changes, introducing the standard
cheats. Your simulated eyes are now smaller than a light
wave, making focus impossible, but the goggles snap your
vision into sharpness and show the atoms around you as small
spheres. (Real nanomachines
are as blind as you were a moment ago, and can't cheat.) You
are on the surface of the 1990s computer chip, between a cell
and two blocky nanocomputers like the ones in Figure 1D. Your
simulated body is 50 nanometers tall, about 1/40,000,000 your
real size, and the smaller nanocomputer is twice your height.
At that size, you can "see" atoms and molecules, as
in Figure 1E.
The simulation keeps bombarding you with air
molecules, but the standard settings leave out the sensation
of being pelted with marbles. A moment ago you were stuck
tight to the ground by molecular stickiness, but the standard
settings give your muscles the effective strength of
steel—at least in simulation—by making everything
around you much softer and weaker. The tourguide says that
the only unreal features of the simulation have to do with you—not
just your ability to see and to ignore thermal shaking and
bombardment, but also your sheer existence at a size too
small for anything so complex as a human being. It also
explains why you can see things move, something about slowing
down everything around you by a factor of 10 for every factor
of 10 enlargement, and by another factor to allow for your
being made stronger and hence faster. And so, with your
greater strength and some adjustments to make your arms,
legs, and torso less sticky, you can stand, see, feel, and
take stock of the situation.
Molecular Texture
The ground underfoot, like everything around
you, is pebbly with atom-sized bumps the size of your
fingertips. Objects look like bunches of transparent grapes
or fused marbles in a variety of pretty but imaginary colors.
The simulation displays a view of atoms and molecules much
like those used by chemists in the 1980s, but with a sharper
3-D image and a better way to move them and to feel the
forces they exert. Actually, the whole simulation setup is
nothing but an improved version of systems built in the late
1980s—the computer is faster, but it is calculating the
same things. The video goggles are better and the whole-body
powersuit is a major change, but even in the 1980s there were
3-D displays for molecules and crude devices that gave a
sense of touching them.
The gloves on this suit give the sensation of
touching whatever the computer simulates. When you run a
fingertip over the side of the smaller nanocomputer, it feels
odd, hard to describe. It is as if the surface were
magnetic–it pulls on your fingertip if you move close
enough. But the result isn't a sharp click of contact,
because the surface isn't hard like a magnet, but strangely
soft. Touching the surface is like touching a film of fog
that grades smoothly into foam rubber, then hard rubber, then
steel, all within the thickness of a sheet of corrugated
cardboard. Moving sideways, your fingertip feels no texture,
no friction, just smooth bumps more slippery than oil, and a
tendency to get pulled into hollows. Pulling free of the
surface takes a firm tug. The simulation makes your
atom-sized fingertips feel the same forces that an atom
would. It is strange how slippery the surface is—and it
can't have been lubricated, since even a single oil molecule
would be a lump the size of your thumb. This slipperiness
makes it obvious how nano-scale
bearings can work, how the parts of molecular machines
can slide smoothly.
But on top of this, there is a tingling
feeling in your fingers, like the sensation of touching a
working loudspeaker. When you put your ear against the wall
of the nanocomputer, you flinch back: for a moment, you heard
a sound like the hiss of a twentieth—century television
tuned to a channel with no broadcast, with nothing but snow
and static—but loud, painfully loud. All the atoms in
the surface are vibrating at high frequencies, too fast to
see. This is thermal vibration, and it's obvious why it's
also called thermal noise.
Gas and Liquid
Individual molecules still move too quickly
to see. So, to add one more cheat to the simulation, you
issue the command "Whoa!", and everything around
seems to slow down by a factor of ten.
On the surface, you now can see thermal
vibrations that had been too quick to follow. All around, air
molecules become easier to watch. They whiz about as thick as
raindrops in a storm, but they are the size of marbles and
bounce in all directions. They're also sticky in a magnetlike
way, and some are skidding around on the wall of the
nanocomputer. When you grab one, it slips away. Most are like
two fused spheres, but you spot one that is perfectly
round—it is an argon atom, and these are fairly rare.
With a firm grip on all sides to keep it from shooting away
like a watermelon seed, you pinch it between your
steel-strong fingers. It compresses by about 10 percent
before the resistance is more than you can overcome. It
springs back perfectly and instantly when you relax, then
bounces free of your grip. Atoms have an unfamiliar
perfection about them, resilient and unchanging, and they
surround you in thick swarms.
At the base of the wall is a churning blob
that can only be a droplet of water. Scooping up a handful
for a closer look yields a swarm of molecules, hundreds, all
tumbling and bumbling over one another, but clinging in a
coherent mass. As you watch, though, one breaks free of the
liquid and flies off into the freer chaos of the surrounding
air: the water is evaporating. Some slide up your arm and
lodge in the armpit, but eventually skitter away. Getting rid
of all the water molecules takes too much scraping, so you
command "Clean me!" to dry off.
Too Small and Too Large
Beside you, the smaller nanocomputer is a
block twice your height, but it's easy to climb up onto it as
the tourguide suggests. Gravity is less important on a small
scale: even a fly can defy gravity to walk on a ceiling, and
an ant can lift what would be a truck to us. At a simulated
size of fifty nanometers, gravity counts for nothing.
Materials keep their strength, and are just as hard to bend
or break, but the weight of an object becomes negligible.
Even without the strength-enhancement that lets you overcome
molecular stickiness, you could lift an object with 40
million times your mass–like a person of normal size
lifting a box containing a half-dozen fully loaded oil
tankers. To simulate this weak gravity, the powersuit cradles
your body's weight, making you feel as if you were floating.
This is almost like a vacation in an orbital theme park,
walking with stickyboots on walls, ceilings, and whatnot, but
with no need for antinausea medication.
On top of the nanocomputer is a stray protein
molecule, like the one in Figure 1E. This looks like a
cluster of grapes and is about the same size. It even feels a
bit like a bunch of grapes, soft and loose. The parts don't
fly free like a gas or tumble and wander like a liquid, but
they do quiver like gelatin and sometimes flop or twist. It
is solid enough, but the folded structure is not as strong as
your steel fingers. In the 1990s, people began to build
molecular machinery out of proteins, copying biology. It
worked, but it's easy to see why they moved on to better
materials.
From a simulated pocket, you pull out a
simulated magnifying glass and look at the simulated protein.
This shows a pair of bonded atoms on the surface at 10 times
magnification, looking like Figure 1F. The atoms are almost
transparent, but even a close look doesn't reveal a nucleus
inside, because it's too small to see. It would take 1,000
times magnification to be able to see it, even with the head
start of being able to see atoms with your naked eye. How
could people ever confuse big, plump atoms with tiny specks
like nuclei? Remembering how your steel-strong fingers
couldn't press more than a fraction of the way toward the
nucleus of an argon atom from the air, it's clear why nuclear
fusion is so difficult. In fact, the tourguide said that it
would take a real-world projectile over a hundred times
faster than a high-powered rifle bullet to penetrate into the
atomic core and let two nuclei fuse. Try as you might, there
just isn't anything you could find in the molecular world
that could reach into the middle of an atom to meddle with
its nucleus. You can't touch it and you can't see it, so you
stop squinting though the magnifying glass. Nuclei just
aren't of much interest in nanotechnology.
Puzzle Chains
Taking the advice of the tourguide, you grab
two molecular knobs on the protein and pull. It resists for a
moment, but then a loop comes free, letting other loops flop
around more, and the whole structure seems to melt into a
writhing coil. After a bit of pulling and wrestling, the
protein's structure becomes obvious: It is a long
chain–longer than you are tall, if you could get it
straight—and each segment of the chain has one of
several kinds of knobs sticking off to the side. With the
multicolored, glassy-bead portrayal of atoms, the protein
chain resembles a flamboyant necklace. This may be
decorative, but how does it all go back together? The chain
flops and twists and thrashes, and you pull and push and
twist, but the original tight, solid packing is lost. There
are more ways to go wrong in folding up the chain than there
are in solving Rubik's Cube, and now that the folded
structure is gone, it isn't even clear what the result should
look like. How did those twentieth-century researchers ever
solve the notorious "protein folding problem"? It's
a matter of record that they started building protein objects
in the late 1980s.
This protein molecule won't go back together,
so you try to break it. A firm grip and a powerful yank
straightens a section a bit, but the chain holds together and
snaps back. Though unfolding it was easy, even muscles with
the strength of steel—the strength of
Superman—can't break the chain itself. Chemical bonds
are amazingly strong, so it's time to cheat again. When you
say, "Flimsy world–one second!" while pulling,
your hands easily move apart, splitting the chain in two
before its strength returns to normal. You've forced a
chemical change, but there must be easier ways since chemists
do their work without tiny superhands. While you compare the
broken ends, they thrash around and bump together. The third
time this happens, the chain rejoins, as strong as before.
This is like having snap-together parts, but the snaps are
far stronger than welded steel. Modern assembler chemistry
usually uses other approaches, but seeing this happen makes
the idea of molecular assembly more understandable: Put the
right pieces together in the right positions, and they snap
together to make a bigger structure.
Remembering the "Whoa!" command,
you decide to go back to the properly scaled speed for your
size and strength. Saying "Standard settings!," you
see the thrashing of the protein chain speed up to
hard-to-follow blur.
Nanomachines
At your feet is a ribbed, ringed cylindrical
object about the size of a soup can—not a messy, loosely
folded strand like the protein (before it fell apart), but a
solid piece of modern nanotechnology. It's a gear like the
one in Figure 1E. Picking it up, you can immediately feel how
different it is from a protein. In the gear, everything
is held in place by bonds as strong as those that strung
together the beads of the protein chain. It can't unfold, and
you'd have to cheat again to break its perfect symmetry. Like
those in the wall of the nanocomputer, its solidly attached atoms vibrate only slightly.
There's another gear nearby, so you fit them together and
make the atomic teeth mesh, with bumps on one fitting into
hollows on the other. They stick together, and the soft,
slick atomic surfaces let them roll smoothly.
Underfoot is the nanocomputer itself, a huge
mechanism built in the same rigid style. Climbing down from
it, you can see through the transparent layers of the wall to
watch the inner works. An electric motor an arm-span wide
spins inside, turning a crank that drives a set of
oscillating rods, which in turn drive smaller rods. This
doesn't look like a computer; it looks more like an
engineer's fantasy from the nineteenth century. But then, it
is an antique design–the tourguide said that the
original proposal was a piece of exploratory
engineering dating from the mid-1980s, a mechanical
design that was superseded by improved electronic designs
before anyone had the tools to build even a prototype. This
simulation is based on a version built by a hobbyist many
years later.
The mechanical nanocomputer may be crude, but
it does work, and it's a lot smaller and more efficient than
the electronic computers of the early 1990s. It's even
somewhat faster. The rods slide back and forth in a blur of
motion, blocking and unblocking each other in changing
patterns, weaving patterns of logic. This nanocomputer is a
stripped-down model with almost no memory, useless by itself.
Looking beyond it, you see the other block–the one on
the left in Figure 1D–which contains a machine powerful
enough to compete with most computers built in 1990. This
computer is a millionth of a meter on a side, but from where
you stand, it looks like a blocky building looming over ten
stories tall. The tourguide says that it contains over 100
billion atoms and stores as much data as a room full of
books. You can see some of the storage system inside: row
upon row of racks containing spools of molecular tape
somewhat like the protein chain, but with simple bumps
representing the 1s and 0s of computer data.
These nanocomputers seem big and crude, but
the ground you're now standing on is also a computer–a
single chip from 1990, roughly as powerful as the smaller,
stripped-down nanocomputer at your side. As you gaze out over
the chip, you get a better sense for just how crude things
were a few decades ago. At your feet, on the smallest scale,
the chip is an irregular mess. Although the wall of the
nanocomputer is pebbly with atomic-scale bumps, the bumps are
as regular as tile. The chip's surface, though, is a jumble
of lumps and mounds. This pattern spreads for dozens of paces
in all directions, ending in an irregular cliff marking the
edge of a single transistor. Beyond, you can see other ridges
and plateaus stretching off to the horizon. These form grand,
regular patterns, the circuits of the computer. The
horizon–the edge of the chip–is so distant that
walking there from the center would (as the tourguide warns)
take days. And these vast pieces of landscaping were
considered twentieth-century miracles of miniaturization?
Cells and Bodies
Even back then, research in molecular biology
had revealed the existence of smaller, more perfect machines
such as the protein molecules in cells. A simulated human
cell–put here because earlier visitors wanted to see the
size comparisons–its on the chip next to the smaller
nanocomputer. The tourguide points out that the simulation
cheats a bit at this point, making the cell act as though it
were in a watery environment instead of air. The cell dwarfs
the nanocomputer, sprawling across the chip surface and
rearing into the sky like a small mountain. Walking the
nature trail around its edge would lead across many
transistor-plateaus and take about an hour. A glance is
enough to show how different it is from a nanocomputer or a
gear: it looks organic, it bulges and curves like a
blob of liver, but its surface is shaggy with waving
molecular chains.
Walking up to its edge, you can see that the
membrane wrapping the cell is fluid (cell walls are
for stiff things like plants), and the membrane molecules are
in constant motion. On an impulse, you thrust your arm
through the membrane and poke around inside. You can feel
many proteins bumping and tumbling around in the cell's
interior fluid, and a crisscrossing network of protein cables
and beams. Somewhere inside are the molecular machines that
made all these proteins, but such bits of machinery are
embedded in a roiling, organic mass. When you pull your arm
out, the membrane flows closed behind. The fluid, dynamic
structure of the cell is largely self healing. That's what
let scientists perform experimental surgery on cells with the
old, crude tools of the twentieth century: They didn't need
to stitch up the holes they made when they poked around
inside.
Even a single human cell is huge and complex.
No real thinking being could be as small as you are in the
simulation: A simple computer without any memory is twice
your height, and the larger nanocomputer, the size of an
apartment complex, is no smarter than one of the submoronic
computers of 1990. Not even a bendable finger could be as
small as your simulated fingers: in the simulation, your
fingers are only one atom wide, leaving no room for the
slimmest possible tendon, to say nothing of nerves.
For a last look at the organic world, you
gaze out past the horizon and see the image of your own,
full-sized thumb holding the chip on which you stand. The
bulge of your thumb rises ten times higher than Mount
Everest. Above, filling the sky, is a face looming like the
Earth seen from orbit, gazing down. It is your own face, with
cheeks the size of continents. The eyes are motionless.
Thinking of the tourguide's data, you remember: The
simulation uses the standard mechanical scaling rules, so
being 40 million times smaller has made you 40 million times
faster. To let you pull free of surfaces, it increased your
strength by more than a factor of 100, which increased your
speed by more than a factor of 10. So one second in the
ordinary world corresponds to over 400 million here in the
simulation. It would take years to see that huge face in the
sky complete a single eyeblink.
Enough. At the command "Get me
out!", the molecular world vanishes, and your feeling of
weight returns as the suit goes slack. You strip off the
video goggles—and hugely, slowly, blink.
|